
A Simple Product Roadmap Playbook for Medical Devices.
By Balasubramanian Srinivasan — Responsible AI/GenAI Lead
In the enterprise AI & Connected Medical Device world, a product roadmap is not a feature wish‑list. It is a simple, shared plan to patient safety and regulatory excellence. For Product Managers and QARA leads, "moving fast" must always be balanced with "staying compliant."
This blog page is a starting point that bridges a gap between an idea and reality.
Start With Three Outcomes/Turn Vision into a Structured Roadmap
Before listing features, write down product key performance indicators or KRA(Key result areas) your product must achieve.
Patient and user outcome:
User Needs & Intended use: What should improve or become safer (e.g., fewer errors, faster diagnosis, less training time, 20% fast recovery, customer outcome)?
Regulatory and safety outcome:
Embed Safety First: Start your Risk Management File (ISO 14971) - What level of risk control and compliance do you need in each target market? Start your preliminary hazard analysis as soon as you define the use case, Align features to risk controls, Include cyber‑security and data privacy controls as first‑class items for connected devices and software. Simple thumb rule If you can’t mitigate the Safety and Security risk, the feature shouldn't be on the roadmap.
Business outcome:
What success looks like - Product realization, Market Release" to gather early user feedback.
If a feature does not support at least one of these outcomes, probably is just a wish list not a product roadmap.
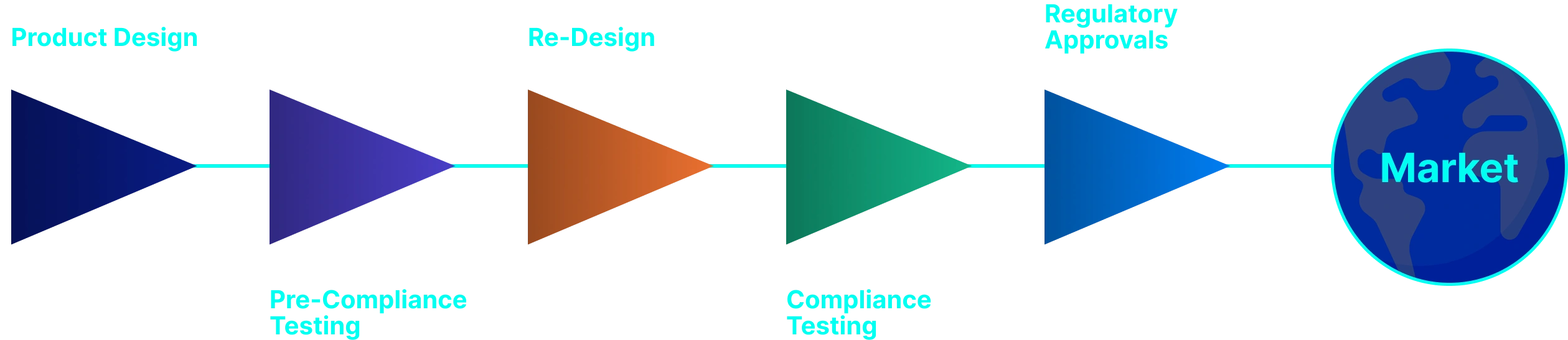
Use Four Simple Phases
Always keep product life cycle view simple and visible to all teams.
Discover & Design
- Clarify the problem, users, and workflows.
- Capture early risks, regulatory path, and UX needs.
Build & Prove
- Develop hardware/software/prototype.
- Run verification, validation, and usability testing.
- Ensure your roadmap includes a phase for moving from prototype to "routine manufacturing" under a certified Quality Management System (ISO 13485).
Approve & Launch
- Start with a "Limited Market Release" to gather early user feedback.
- Prepare regulatory submissions and technical files.
- Get markets ready: training, support, and initial customers.
Post-Market Surveillance (PMS) & Continuous Improvement
- Collect complaints, feedback, and real‑world data.
- Prioritize fixes, enhancements, and new indications.
Always keep a roadmap as living documents stay honest, current, and responsible.
Revisit and adjust quarterly (or more often for software and SaMD), with explicit decisions on what moves, what drops, and why.
Establish AI Governance practices cross‑functional reviews that include product, engineering, QARA, clinical, legal and commercial stakeholders.
Validate all clinical and safety‑related insights with appropriate experts and evidence.
Be transparent about the limits of your models and data.
Embed Safety into your roadmap, ensure privacy, security, and fairness requirements are treated as non‑negotiable constraints, not optional features.
How different roles use the roadmap
| Role | What they need from the roadmap |
|---|---|
| Product managers | Outcomes, priorities, trade‑offs, and cross‑functional dependencies. |
| QA/RA leaders | Safety and regulatory milestones, evidence plans, and documentation timelines. |
| UI/UX designers | User research schedule, usability test waves, and UX‑impacting releases. |
| Product engineers | Clear sequence of design, V&V, and sustaining work. |
| Product management consultants | Portfolio‑level view, market entry waves, and positioning evolution. |
Source References:
- ISO 13485:2016 – Medical Devices Quality Management Systems.
- FDA Design Control Guidance (21 CFR 820.30).
- EU Medical Device Regulation (MDR 2017/745).
Conclusion
Contact our subject matter expert bala@cdaqi.com for free 30mins consultation to help your team.

