EMI/EMC Testing Service
Our certifications demonstrate that your products have been tested to applicable standards. We are recognized regulatory expertise, who provide critical credibility to authorities and the marketplace. Bringing your products to market more efficiently and clearly to differentiate from crowded shelves with certification from our Team
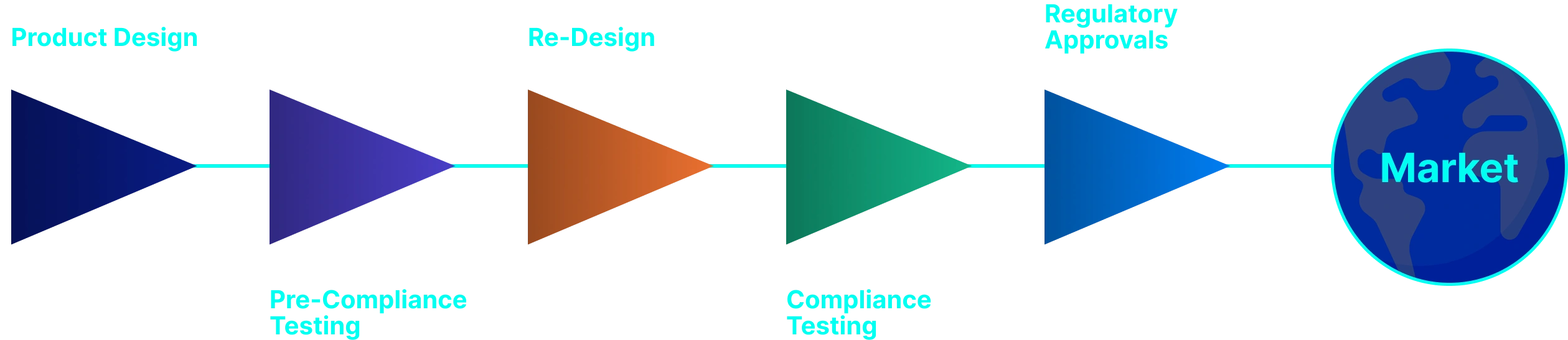
EMI/EMC Testing Implementation Approach
- Including: Kick-off + Discovery + Scope & Deliverables
- Align on goals, target markets, timelines, and applicable standards (IEC 60601-1, IEC 60601-1-2, etc.).
- Collect technical data, review risks, and finalize the test plan, scope, schedule, and commercial proposal with client sign-off
- Perform pre-compliance and formal compliance tests for electrical safety and EMC as per the agreed plan.
- Support issue debugging/re-design and provide interim updates so the manufacturer can make timely design decisions.
- Issue final test reports, conformance statements, and any certificates needed for regulatory submissions.
- Conduct a closing review meeting to explain results, recommend next steps, and support documentation for approvals and market launch.
EMI/EMC Testing Service Process
Electrical and Electromegnetic compatibility test for regulatory submissions
Medical Devices must meet Strict global standards for electrical safety and electromagnetic compatibility to ensure they are safe and do not interfere with other critical components.
Electrical safety test readiness insights
EMI/EMC Testing Services - Key Benefits
Key Benefits
- Regulatory Compliance and Global Access: Testing to standards like IEC 60601-1 and IEC 60601-1-2 supports CE marking, FDA approvals, and international market entry by verifying safety against electrical hazards and electromagnetic interference.
- Risk Reduction and Cost Savings: Early detection of issues minimizes recalls, liability, rework, and downtime while extending equipment lifespan through preventive maintenance.


Why Our Structured Approach Matters
This tailored approach directly support our core differentiator the integrated medical device electrical safety standard to align on goals, target markets and delivery timelines, test reports needed for regulatory submissions.
Key Recommendation for
Manufacturers


Get a tailored readiness plan in 14 days - with prioritized fixes and regulatory mapping.